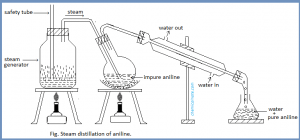
In Hoffmann’s method, the given mixture of primary, secondary and tertiary amines is heated with diethyl oxalate when,
1) 10 amine forms dialkyl oxamide which is crystalline solid.

2) 20 amine forms dialkyl oxamic ester which is an oily liquid.

3) 30 amine does not react as it does not contain replaceable hydrogen atom on nitrogen.
The reaction mixture containing dialkyl oxamide, dialkyl oxamic ester, tertiary amine and ethyl alcohol is first filtered and solid product of dialkyl oxamide is separated. Dialkyl oxamide is heated with aq.KOH to recover primary amine.

The remaining mixture of dialkyl oxamic ester, tertiary amine and ethyl alcohol is subjected to fractional distillation. Tertiary amine is distilled out first. The residual dialkyl oxamic ester is heated with aq. KOH to recover secondary amine and alcohol in different fractions.

In this way, the given mixture of 10,20 and 30 amines are separated by Hoffmann’s method.
References
- Sthapit, M.K., Pradhananga, R.R., Foundations of Chemistry, Vol 1 and 2, Fourth edition, Taleju Prakashan, 2005.
- Ghosh, S.K., Advanced General Organic Chemistry, Second Edition, New Central Book Agency Pvt. Ltd., Kolkatta, 2007.
- Morrison, R.T. , Boyd, R.N., Organic Chemistry, Sixth edition, Prentice-Hall of India Pvt. Ltd., 2008.
- https://chemicalnote.com/aniline-lab-preparation-properties-reactions-and-uses/