Contents
Laboratory preparation of chloroform (trichloromethane) (CHCl3)
Principle:
Chloroform is prepared in the laboratory by heating ethanol or acetone with aqueous bleaching powder paste. Bleaching powder paste acts as oxidizing, chlorinating and hydrolyzing agent.

From ethanol :
Step I : Oxidation :

Step II : Chlorination :

Step III : Hydrolysis :

From acetone (propanone) :
Step I : Chlorination :

Step II : Hydrolysis :

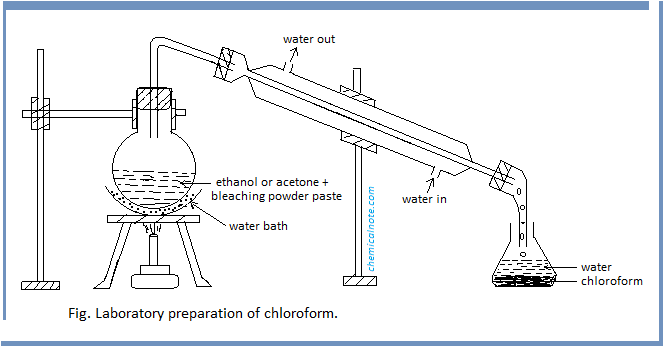
Procedure : First of all, bleaching powder paste is prepared by mixing 100 gm of bleaching powder with 200 ml of water in one liter round bottomed flask and 25ml of ethanol or acetone is added to it. The flask is heated gently on a water bath until a mixture of chloroform and water distils over, as shown in figure. The mixture from receiver is transferred into a separating funnel and the lower layer of chloroform is separated.
Purification : The impure chloroform is washed with dilute caustic soda (NaOH) solution and then with water successively in the separating funnel. It is then dried over anhydrous calcium chloride and redistilled between 60 -650C. In this way, pure and dry chloroform is obtained.
See the properties of chloroform…….
Laboratory preparation of diethyl ether (ethoxyethane)
When an excess of ethyl alcohol is heated with conc. H2SO4 at 1400C, diethyl ether is obtained.
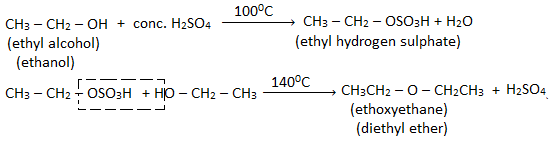

Procedure: A mixture of ethyl alcohol and conc. H2SO4 in the ratio of 1:1 by volume is taken in the distillation flask. The flask is then fitted with a dropping funnel containing alcohol. The flask is heated on a sand bath at 1400C. Ethanol is added at nearly the same rate that of the distillation so that the ether formed is continuously received in the receiver kept cold in the ice cold water.
Purification: The distillate thus obtained contains ether, ethyl alcohol, water and sulphurous acid. The acid is removed by washing with KOH or NaOH solution. The solution is then stirred with anhydrous CaCl2 to remove alcohol. Finally it is redistilled to obtain almost pure ether.
See the properties of diethyl ether……
Laboratory Preparation of Nitrobenzene
It is prepared in lab by heating benzene with conc. HNO3 and conc. H2SO4 at 600C.


Procedure : 50 ml of benzene is taken in a round bottomed flask. To this flask, 60 ml conc. HNO3 and 60 ml conc. H2SO4 (i.e. nitrating mixture) is added a little at a time, shaking and cooling after each addition. Then the mixture is heated (refluxed) in water bath at 600C for about one and half hour till the yellow oily layer appears on the surface. The flask is then cooled and the layer of nitrobenzene is separated by using separating funnel.

Purification: It is first washed with dil. Na2CO3 to remove the acidic impurities and then with water several times. It is then dried over fused calcium chloride. It is finally distilled at 2110C to get pure nitrobenzene.
See the properties of nitrobenzene….
Laboratory preparation of aniline
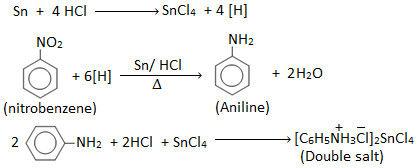
Aniline is prepared in laboratory by reducing nitrobenzene with tin (Sn) and conc. HCl.
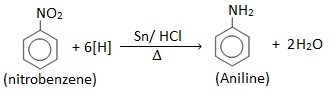

Procedure: 10 ml nitrobenzene and 20 gm of granulated tin are placed in the 250 ml round bottom flask fitted with a reflux condenser. 50 ml of conc. HCl is added gradually with constant shaking. After each addition, the round bottom flask is cooled so that temperature may not go above 900C. Then the reaction mixture is heated on a boiling water bath for about one hour until the reaction is completed which is indicated by the smell of nitrobenzene, the disappearance of smell indicates the completion of the reaction. The flask is then cooled and a crystalline solid mass of double salt is separated out.
The crystalline solid mass is then treated with conc. NaOH until the solution is cleared and becomes strongly alkaline. Aniline is separated out and is floated on the surface as dark brown oil.
![]()
The mixture obtained is then subjected to the process of steam distillation until clear distillate is obtained.

Purification: Aniline is extracted by shaking the distillate several times with ether. The ethereal layer is separated each time with the help of separating funnel. Now, the ethereal aniline is placed for the evaporation where ether evaporates out. Aniline thus obtained is finally purified by redistillation at 182-1840C.
See the properties of aniline…..
Laboratory preparation of anhydrous formic acid (methanoic acid)
Formic acid is prepared in the laboratory by the decarboxylation of oxalic acid with glycerol at 1100C.

This reaction occurs in following steps:

Procedure: About 50ml of anhydrous glycerol and 40gm of oxalic acid crystals are placed in a flask and all the apparatus are fitted as shown in figure. The flask is heated at 1100C till the evolution of carbon dioxide (Marked by effervescence) ceases. The reaction flask is then cooled and a fresh lot of oxalic acid (40gm) is added. The mixture is again heated at 1100C and the distilled aqueous solution of formic acid is collected in the receiver.

For anhydrous formic acid:
The aqueous formic acid is neutralized with lead carbonate and the lead formate solution is then evaporated to get crystals of lead formate. The dry lead formate is packed in the inner tube of condenser and treated with dry H2S gas. As a result anhydrous formic acid is formed which is collected in the receiver.

Anhydrous formic acid so obtained contains traces of H2S. it is mixed with some lead formate and is distilled to obtain pure formic acid.

Note: Anhydrous HCOOH can’t be obtained by distillation of aqueous HCOOH as b.pt. of HCOOH (b.pt. 100.50C) is almost similar to that of water.
See the properties of formic acid……
References
- Bahl, B.S., A., Advanced Organic Chemistry, S. Chand and company Ltd, New Delhi, 1992.
- Finar, I. L., Organic Chemistry, Vol. I and Vol. II, Prentice Hall, London, 1995.
- Ghosh, S.K., Advanced General Organic Chemistry, Second Edition, New Central Book Agency Pvt. Ltd., Kolkatta, 2007.
- Morrison, R.T. , Boyd, R.N., Organic Chemistry, Sixth edition, Prentice-Hall of India Pvt. Ltd., 2008.
- March, j., Advanced Organic Chemistry, Fourth edition, Wiley Eastern Ltd. India, 2005.
- https://chemicalnote.com/ether-isomers-preparation-properties-and-uses/
- https://chemicalnote.com/formic-acid-methanoic-acid-laboratory-preparation-properties-and-uses/
- https://chemicalnote.com/chloroform-lab-preparation-properties-uses-and-question-answer/
- https://chemicalnote.com/aniline-lab-preparation-properties-reactions-and-uses/
- https://chemicalnote.com/nitrobenzene-laboratory-preparation-properties-and-uses/



