Contents
What are aromatic aldehydes and ketones?
Aromatic aldehydes are the compounds in which –CHO group is bonded directly to an aromatic ring. Eg.

Aromatic ketones are the compounds in which carbonyl group is bonded with either both aryl group or aryl and alkyl group. Eg.

Note: The compound in which carbonyl group is not bonded directly to the benzene ring are considered as arly substituted aliphatic aldehydes. Eg.

Preparation of benzaldehyde and acetophenone
Preparation of benzaldehyde:
1. From toluene: Benzaldehyde is prepared by oxidation of toluene with cerium oxide (CeO2) in the presence of conc. H2SO4.

2. From Rosenmund’s reduction : Benzaldehyde is obtained by reducing benzoyl chloride with hydrogen in the presence of Pd catalyst deposited in BaSO4.

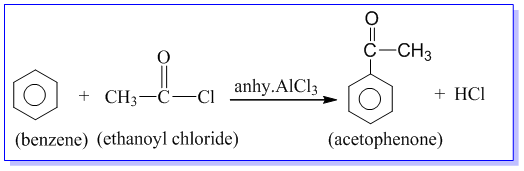
Preparation of acetophenone from benzene:
Acetophenone is prepared by the treatment of benzene with acetyl chloride in the presence of anhydrous AlCl3.
Properties of benzaldehyde
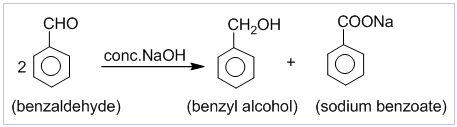
1. Cannizzaro’s reaction:
Aldehydes which do not contain α-hydrogen like HCHO, C6H5CHO,etc. undergo self oxidation and reduction on treatment with conc. alkali. In this reaction one molecule is oxidized to carboxylic acid and other molecule is reduced to alcohol. Thus, a mixture of an alcohol and a salt of carboxylic acid is formed by Cannizzaro’s reaction.
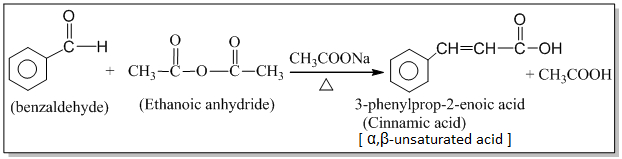
2. Perkin’s (condensation) reaction:
The condensation of an aromatic aldehyde with an acid anhydride in the presence of sodium or potassium salt of the same acid to produce α,β-unsaturated acid is known as the Perkin’s condensation.
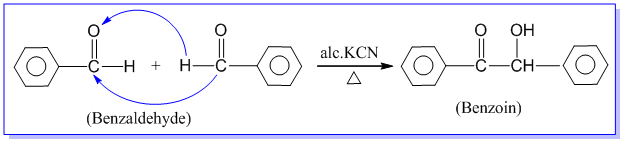
3. Benzoin condensation reaction:
Benzaldehyde when heated with alcoholic solution of potassium cyanide, undergoes self condensation between two molecules to form an α-hydroxy ketone known as benzoin. This reaction is called benzoin condensation reaction. Eg.
4. Electrophilic substitution reaction:
-CHO group is electron withdrawing group. It withdraws ∏- electrons from benzene ring, decreasing electron density of aromatic ring.
Benzaldehyde is resonance hybrid of following resonance structures:
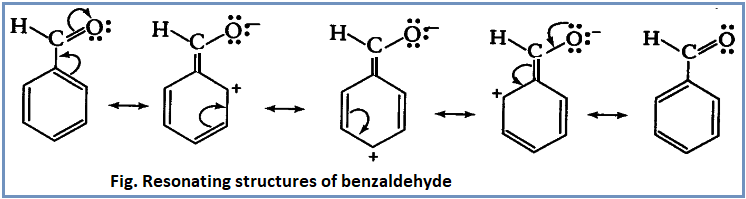
From the above resonating structures, it is clear that electron density is comparatively high at meta position. Thus incoming electrophile attacks at meta position to give meta substituted product. Thus –CHO is a meta directing group.

Similarly, acetophenone also undergoes electrophilic substitution reaction at meta position.

- Other reactions of aromatic aldehydes and ketones are similar to the reactions of aliphatic aldehydes and ketones.
- See the reactions of aliphatic aldehydes and ketones….
EXERCISE
1. An aromatic compound “A’ (Molecular formula C8H8O) gives a positive 2, 4-DNPH test. It gives a yellow precipitate of compound ‘B’ on treatment with iodine and sodium hydroxide solution. Compound ‘A’ does not give Tollen’s or Fehling’s test. On severe oxidation with potassium permanganate forms a carboxylic acid ‘C’ (Molecular formula C7H602), which is also formed along with the yellow compound in the above reaction. Identify A, B and C and write all the reactions involved.
2. An organic compound A, molecular formula C9H10O forms 2,4-DNP derivative, reduces Tollen’s reagent and undergoes Cannizzaro reaction. On vigorous oxidation, it gives 1,2-benzene dicarboxylic acids. Identify A.
[Ans: The compound A will be 2-Ethylbenzaldehyde]
3. A liquid of molecular formula C7H6O forms an oxime, reduces Tollen’s reagent and undergoes Clemmensen reduction to give toluene. Explain the reaction involved and write the structural formula of this liquid.
4. Compound (A) C6H12 decolorizes bromine, but gives no reaction with sodium metal or phenylhydrazine. Ozonolysis of (A) gives two compounds, (B) and (C), both react with phenylhydrazine. Compound (B) gives positive Tollens’ and iodoform tests. Compound (C) has molecular weight of 72. Suggest structures for (A), (B) and (C). Give equations for all reactions.
5. An organic compound (A) which has characteristic odour, on treatment with NaOH forms two compounds (B) and (C). Compound (B) has the molecular formula C7H8O which on oxidation with CrO3 gives back compound (A). Compound (C) is sodium salt of the acid. Compound (C) when heated with soda lime yields an aromatic hydrocarbon (D). Deduce the structures of (A), (B), (C) and (D). Write chemical equations for all reactions taking place.
6. Why are aromatic carbonyl compounds less reactive than those of aliphatic ones?
7. Name two carbonyl compounds which do not produce crystalline products with sodium bisulphate.
[Ans. Diethyl ketone, acetophenone, benzophenone etc. do not undergo reaction with NaHSO3. It is due to steric hindrance of the bulky groups present around the carbonyl group.]
8. Why is –CHO group meta directing towards electrophilic substitution in aromatic aldehyde?
9. Benzaldehyde and acetophenone are two aromatic carbonyl compounds.
a. Write any two methods of preparation of benzaldehyde.
b. Write any two methods of preparation of acetophenone.
c. Which compound give iodoform test? Give chemical reaction.
Convert:
- Nitrobenzene into acetophenone.
- Benzaldehyde to cinnamic acid
Write short note on:
- Perkin’s condensation reaction
- Benzoin condensation reaction
- Cannizzaro’s reaction
- DNPH test
How do you distinguish:
- Benzaldehyde from acetophenone
- Acetophenone from benzophenone
What happens when:
- Benzaldehyde is heated with 50% NaOH.
- Benzaldehyde is heated with aqueous ethanolic KCN solution.
- Benzaldehyde is heated with ethanoic anhydride in presence of sodium acetate.
- Toluene is oxidized with CeO2 in presence of conc. H2SO4.
- Benzaldehyde is heated with lithium aluminum hydride.
- Benzaldehyde is nitrated.
REFERENCES
- Ghosh, S.K., Advanced General Organic Chemistry, Second Edition, New Central Book Agency Pvt. Ltd., Kolkatta, 2007.
- Morrison, R.T. , Boyd, R.N., Organic Chemistry, Sixth edition, Prentice-Hall of India Pvt. Ltd., 2008.
- https://www.snapsolve.com/class11/chemistry/cbse-1100177433
- https://chemicalnote.com/aldehydes-and-ketones-carbonyl-compounds-preparation-and-properties/



