Test reactions can be used to distinguish between different organic compounds. Test reactions give a visual clue (i.e. colour change, precipitate, or gas formation) as evidence for a chemical reaction.
In this note, we have describes about five test reactions – iodoform test, silver mirror test, DNP test, carbylamines test and nitrous acid test.

Contents
Iodoform test

This reaction occurs in same way as lab preparation of chloroform. Eg.
- This reaction is used to distinguish some of the pairs of compounds. Eg.
- Ethanol and methanol
- Ethanal and methanal
- 2-pentanone and 3-pentanone, etc.
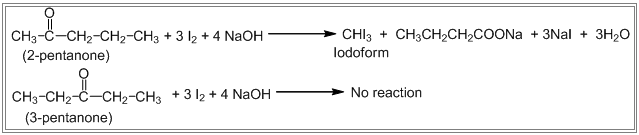
Q) How can you distinguish 2-pentanone and 3-pentanone.
2-pentanone and 3-pentanone can be distinguished by iodoform test as 2-pentanone gives iodoform reaction but 3-pentanone doesn’t give iodoform reaction. Eg.
Tollen’s test [Silver mirror test]
Tollen’s reagent is an ammonical solution of silver nitrate. It is prepared by adding dilute solution of NH4OH to AgNO3 solution till the precipitate of Ag2O once formed gets dissolved.

Aldehydes on heating with Tollen’s reagent reduces the reagent to metallic silver.
![Tollen’s test [Silver mirror test]](https://chemicalnote.com/wp-content/uploads/2021/05/word-image-69.png)
The silver deposits on the inner wall of test tube forming a shining layer like mirror. Hence, this test is known as silver mirror test.
- Both aliphatic and aromatic aldehydes give this test but ketones do not give this test.
Q) Write the functional isomer of C3H6O and give a chemical test to distinguish them.
The functional isomers of C3H6O are:

These two isomers can be distinguished by silver mirror test (i.e. Tollen’s reagent). Propanal (i.e. aldehyde) gives positive silver mirror test but propanone (i.e. ketone) does not give this test.

Note: Formic acid (methanoic acid) possesses dual functional groups i.e. carboxylic group as well as aldehyde group. Hence, formic acid also gives positive silver mirror test but other carboxylic acids (like acetic acid) do not give this test.

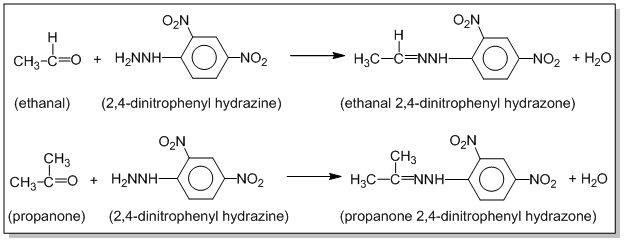
2,4-DNP test
Aldehydes and ketones react with 2,4-dinitrophenyl hydrazine (2,4-DNP) to form yellow, orange or red ppt. of 2,4-dinitrophenyl hydrazone. Eg.
Q) Write a chemical test to distinguish ethanal from ethanol?
Ethanal and ethanol can be distinguished by 2,4-DNP test. Ethanal (i.e aldehyde) reacts with 2,4-dinitrophenyl hydrazine (2,4-DNP) to form orange ppt. of ethanal 2,4-dinitrophenyl hydrazone. But ethanol does not give this test.

Note : 2,4-DNP = Brady’s reagent.
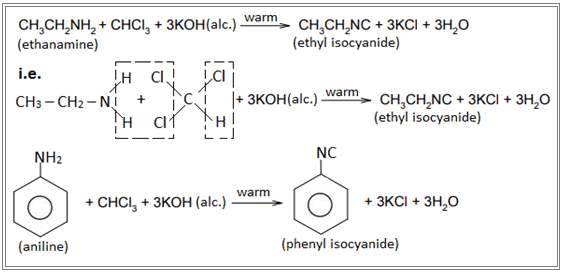
Carbylamine test : Test reaction of primary amines
When primary amine is warmed with chloroform in the presence of alcoholic KOH, an offensive (unpleasant) smell of carbylamines ( i.e. isocyanide) is obtained. This reaction is known as carbylamine reaction. Eg.
Secondary and tertiary amines do not respond to this reaction and therefore, this reaction is used as test reaction for primary amines.
Q) How would you distinguish ethanamine and N-ethylethanamine ?
Ethanamine and N-methylmethanamine can be distinguished by carbylamines test. Ethanamine (i.e. primary amine) reacts with chloroform and alcoholic KOH to form ethyl isocyanide having an offensive smell. But N-methylmethanamine (i.e. secondary amine) does not give this test.

Nitrous acid test- Test reaction of 10, 20 and 30 amines
Primary, secondary and tertiary amines react differently with nitrous acid.
i. Primary amines react with nitrous acid to produce nitrogen gas which is seen as bubbles.


ii. Secondary amines react with nitrous acid to produce a yellow oily layer of N-nitrosoamine.

iii. Tertiary amines react with nitrous acid to form soluble trialkyl ammonium nitrite salt. There is no visible sign of reaction.

Q) Give a chemical reaction to distinguish ethanamine from N-methyl methanamine.
Nitrous acid test can distinguish ethanamine from N-methylmethanamine.
i. Ethanamine (i.e. primary amine) reacts with nitrous acid to produce nitrogen gas which is seen as bubbles.
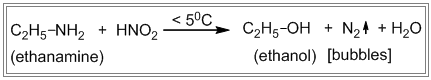
ii. N-methyl methanamine (i.e. secondary amine) reacts with nitrous acid to produce a yellow oily layer of N-nitrosoamine.

Practice questions
Q) How can you distinguish ethanal and methanal.
Q) How can you distinguish 2-pentanone and 3-pentanone.
Q) How can you distinguish propanal and propanone.
Q) Write the functional isomer of C4H8O and give a chemical test to distinguish them.
Q) Write a chemical test to distinguish ethanal from ethanol?
Q) How would you distinguish ethanamine and N,N-diethylethanamine ?
Q) Give a chemical reaction to distinguish 20 and 30 amines having molecular formula C3H9N .
Q) Write a chemical reaction to distinguish formic acid and acetic acid.
References
- Finar, I. L., Organic Chemistry, Vol. I and Vol. II, Prentice Hall, London, 1995.
- Ghosh, S.K., Advanced General Organic Chemistry, Second Edition, New Central Book Agency Pvt. Ltd., Kolkatta, 2007.
- Morrison, R.T. , Boyd, R.N., Organic Chemistry, Sixth edition, Prentice-Hall of India Pvt. Ltd., 2008.
- March, j., Advanced Organic Chemistry, Fourth edition, Wiley Eastern Ltd. India, 2005.
- https://www.jove.com/science-education/11223/identification-of-unknown-aldehydes-and-ketones
- https://www.sciencedirect.com/topics/chemistry/2-4-dinitrophenylhydrazine
- https://www.coursehero.com/file/p71ar0v/Iodoform-test-In-this-test-Acetaldehyde-acetone-and-acetophenone-were-able-to/