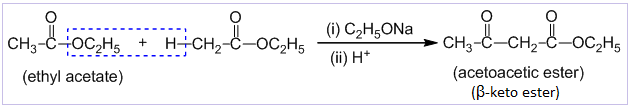
Claisen condensation reaction
Condensation between two molecules of esters having α-hydrogen atom in the presence of strong base like sodium ethoxide and an acid to form β-keto ester is known as Claisen condensation reaction. Eg.
Mechanism of Claisen condensation reaction
Taking ethyl acetate (ethyl ethanoate) as an example, Claisen condensation reaction involves following steps:
Step-I: In this step, ethoxide ion removes a proton from α-carbon of one molecule of ethyl acetate to form a carbocation.
- This step is similar to aldol condensation reaction.

Step-II: In this step, the carbanion (i.e. nucleophile) attacks the carbonyl carbon of second ester molecule to displace ethoxide ion and forms the keto ester.
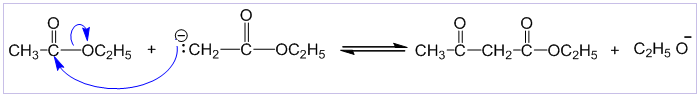
- The claisen condensation differs from aldol condensation at this point. In the aldol condensation, nucleophilic attack leads to addition and in the claisen condensation reaction it leads to substitution.
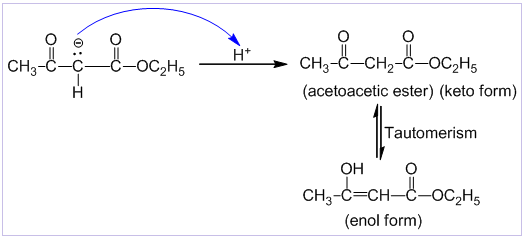
Step-III: In this step, there is an acid base reaction between ethoxide ion and β-keto ester to give β-keto ester anion and ethanol.

Step-IV: In this step, an acid is added to the reaction mixture which gives proton to the β-keto ester anion to form the β-keto ester which is in equilibrium with its enol form.
Note:
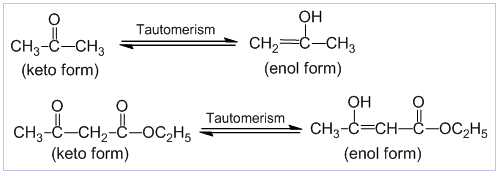
Keto-enol tautomerism :
Keto-enol tautomerism is a special type of isomerism in which two isomeric forms, keto and enol exist in dynamic equilibrium with each other. There is transfer of a α- hydrogen to the oxygen atom of carbonyl group and vice-versa.
References
- Ghosh, S.K., Advanced General Organic Chemistry, Second Edition, New Central Book Agency Pvt. Ltd., Kolkatta, 2007.
- Morrison, R.T. , Boyd, R.N., Organic Chemistry, Sixth edition, Prentice-Hall of India Pvt. Ltd., 2008.
- March, j., Advanced Organic Chemistry, Fourth edition, Wiley Eastern Ltd. India, 2005.
- https://chemicalnote.com/cannizzaros-reaction-and-crossed-cannizzaros-reaction-definition-examples-and-mechanism/
- https://chemicalnote.com/aldol-condensation-and-crossed-aldol-condensation-reaction-definition-examples-mechanism-and-uses/



