Contents
What is ozone and ozone layer?
Ozone is a tri-atomic molecule of oxygen having molecular formula O3 and molecular weight 48.
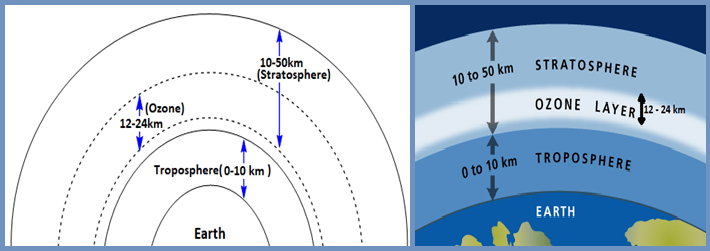
There is a layer of ozone in the atmosphere from 12 to 24 km from the surface of the earth. Ozone is formed naturally in the stratosphere by the action of ultraviolet radiation from the sun on oxygen.

Ozone layer absorbs ultraviolet radiations from the sun and protects the living beings on the earth from the harmful effect of ultraviolet radiation.
Preparation of ozone from oxygen
Ozone is prepared by passing silent electric discharge through pure and dry oxygen in an apparatus called ozonizer. The produced gas is called ozonized oxygen that contains 10-15% ozone.

Structure of ozone
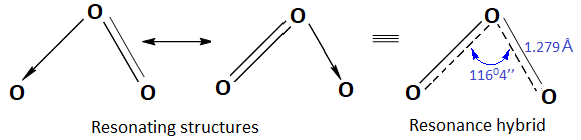
Experimental evidences (like electron diffraction, microwave studies) indicate that ozone molecule is angular with bond angle 11604’’. The bond lengths are equal (1.279 Å ). This indicates ozone molecule is resonance hybrid of structure (I) and (II).
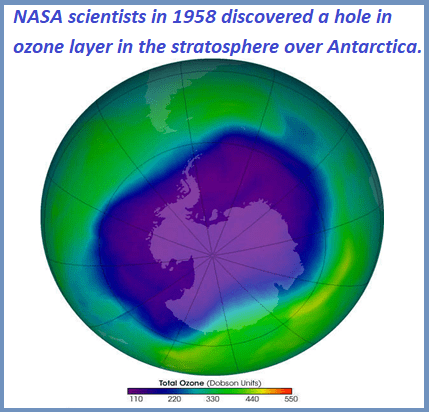
Ozone layer depletion (ozone hole)
Ozone hole means a severe depletion of ozone in the region of ozone layer through which UV rays from the sun can penetrate to the earth causing devastating effects.
Causes of ozone layer depletion :
The main causes responsible for ozone layer depletion are as follows:
Chlorofluorocarbons :
The main cause of ozone layer depletion is manmade chlorofluorocarbons (CFCs). CFCs also known as Freons are compounds containing chlorine, fluorine and carbons. They are extensively used by human beings as coolants in refrigerator, air conditioner etc.
The common CFC’s are CFCl3 (Freon 11), CCl2F2 (Freon 12), CHClF2 (Freon 22) etc.
Because of high stability, freons remain in the environment for a long time and reaches to the stratosphere. In stratosphere, CFCs absorbs UV light and decomposes to give chlorine atom which destroys the ozone molecules by chain reaction as follows:

It is estimated that one molecule of CFC can destroy 100,000 molecules of ozone. Therefore, the use of CFC’S is banned nowadays.
Unregulated rocket launches and nuclear tests :
Researchers say that the unregulated launching of rockets and nuclear tests result in much more depletion of ozone layer than the CFCs do. If not controlled, this might result in a huge loss of the ozone layer.
Nitrogenous Compounds :
The nitrogenous compounds such as NO2, NO, N2O are highly responsible for the depletion of the ozone layer.
Cleaning products and fire extinguishers :
Cleaning products and fire extinguishers contain or produce ozone depleting substances like halons (eg. Bromotrifluoromethane – CBrF3), carbon tetrachloride, hydrofluorocarbons (organic compounds containing hydrogen and fluorine eg. CHF3), etc.
Natural Causes :
The ozone layer has been found to be depleted by certain natural processes such as stratospheric winds. But it does not cause more than 1-2% of the ozone layer depletion.
The volcanic eruptions are also responsible for the depletion of the ozone layer.
Effects of ozone layer depletion:
- Direct exposure to ultraviolet radiations causes health problems such as skin and eye cancer, weekend immune system, etc. in human and other animals.
- Strong ultraviolet rays may lead to minimal growth, flowering and photosynthesis in plants.
- Synthetic polymers, naturally occurring biopolymers, as well as some other materials of commercial interest are adversely affected by UV radiation.
Control measures of ozone layer depletion:
Some points that would help to prevent ozone layer depletion are as follows:
- Avoid the use of dangerous gases like CFCs (chlorofluorocarbons), halogenated hydrocarbons, nitrogen oxides, etc.
- Minimize the use of vehicles – The vehicles emit a large amount of green house gases that lead to the ozone layer depletion. Therefore, the use of vehicles should be minimized as much as possible. Promote the use of electric vehicles and bicycles should be promoted.
- Do not use cleaning products containing halogens (eg.CHF3). We can replace these dangerous substances with safe compounds like bicarbonates.
- Maintain air conditioners, as their malfunctions cause chlorofluorocarbons to escape into the atmosphere.
- Rocket lunching and nuclear tests should be regulated .
Test of ozone
These two reactions can help to identify ozone:
Action with mercury (Mercury tailing) :
When mercury comes in contact with ozone, ozone oxidizes mercury to mercurous oxide. The meniscus of mercury is lowered and it leaves a tail when allowed to fall through an inclined surface. This property of mercury is called tailing of mercury.

Action with KI and starch :
When ozone is passed into the solution of KI and starch, ozone oxidizes KI to iodine and iodine gives blue colour with starch.

These reactions can be used for the test of ozone.
Uses of ozone
- It is used as germicide and disinfectant for the sterilization of water due to its oxidizing nature.
![]()
- It is used to prepare organic compounds like aldehydes and ketones.
- It is used for bleaching delicate articles like silk, starch, wax, etc.
![]()
- It is used for air purification at the crowded places like cinema halls and tunnel railways.

Objective Questions
1. O2 and O3 are:
a. Isotopes c. Allotropes
b. Isobars d. Isomers
2. Ozone is tested by:
a. Ag c. Hg
b. Au d. Zn
3. Ozone is in high concentration in :
a. Troposphere c. Mesosphere
b. Stratosphere d. Thermosphere
4. Tailing of mercury is caused by :
a. Cl2 c. O2
b. H2 d. O3
5. One molecule of chlorofluorocarbons can destroy upto ____ molecules of ozone.
a. 100 c. 10000
b. 1000 d. 100000
6. Montreal protocol is related to the:
a. Global warming
b. Ozone layer depletion
c. Sustainable development
d. Food security
7. Which of the following is known as freon?
a. CCl2F2 c. CF4
b. CHCl3 d. CHF2
Answers :
1.c 2. c 3. b 4. d
5. d 6. b 7. a
References
- Agrawal, S. K., Lal, K., Advanced Inorganic Chemistry, Fifth Revised Edition, Pragati Prakashan, Meerut, 2001.
- Cotton, F. A., and Wilkinson, G., Advanced Inorganic Chemistry, Fifth edition, John Wily and Sons, Singapore, 1995.
- Day, C.M., Selbin, J., Theoritical inorganic Chemistry, second edition, Affiliated East-West Press Pvt. Ltd., New Delhi, 2002.
- https://www.sizes.com/units/dobson_unit.htm#:~:text=A%20unit%20expressing%20the%20concentration,It%20is%20named%20for%20G.%20M.%20B.
- https://www.epa.gov/ozone-pollution-and-your-patients-health/what-ozone#:~:text=Ozone%20(O3)%20is%20a%20highly,either%20good%20or%20bad%20ways.
- https://www.epa.gov/ground-level-ozone-pollution/ground-level-ozone-basics