Besides carbon, hydrogen and oxygen, organic samples may also contain other elements like N, S, halogens, etc. known as foreign elements. These elements can be detected by Lassaigne’s test (method). In this method, little organic compound is fused with sodium metal in a fusion (ignition) tube at high temperature and extracted in distilled water. The resulting aqueous solution is called sodium extract or Lassaigne’s extract.
Contents
Purpose of preparing sodium extract
Organic compounds are covalent compounds. Common laboratory reagents are ionic compounds and handled as aqueous solution. Hence, organic compounds do not interact (react) with common laboratory reagents easily. For example,

When fused with highly reactive sodium metal, at high temperature, covalently bonded atoms of the organic compounds are converted into ionic compounds and can be detected by simple chemical tests using common laboratory reagents.
On fusion following ionic compounds are formed:
If nitrogen is present in organic compounds,

If sulphur is present in organic compounds,

If nitrogen and sulphur both are present together in organic compounds,

If halogens are present in organic compounds,

Preparation of sodium extract
Small piece of dry sodium metal is taken in a fusion tube (ignition tube) and sodium metal is melted and cooled. To this cooled Na- metal, a pinch of organic sample is added then fusion tube is further heated up to red hot. This red hot tube is plunged into distilled water in a porcelain basin. The content is grinded, boiled and filtered to obtain clear filtrate which is known as Na- extract or Lassaigne’s extract.

Q) Sodium extract is alkaline in nature, why?
Sodium metal used during preparation of sodium extract isn’t totally utilized (consumed) by foreign elements and remaining sodium metal combines with water forming sodium hydroxide. Sodium metal combines with water forming sodium hydroxide. Therefore, sodium extract is alkaline in nature.
![]()
Q) Organic compounds are combustible, why?
Organic compounds contain covalent bond which are difficult to break at ordinary temperature. They need huge amount of energy to break and get combusted in presence of oxygen. But once the combustion starts, it produces such a huge amount of energy that the combustion becomes spontaneous i.e. the energy needed is continuously provided by the energy produced. So, organic compounds are highly combustible.
Detection of Nitrogen (N)
If organic sample contain nitrogen, sodium extract has following composition :
![]()
If this sodium extract is not alkaline, make it alkaline by adding dil.NaOH then 1 or 2 drop of freshly prepared FeSO4 solution is added. The content is boiled, cooled and acidified with dil.HCl . Finally 1, 2 drops of ferric chloride (FeCl3) is added. Appearance of prussian blue or green colour indicates the presence of nitrogen in organic compound.

Note : The role of HCl is to dissolve dirty green ppt. of ferrous hydroxide which otherwise masks the colour.
Detection of sulphur
If organic sample contains sulphur, sodium extract has following composition :
![]()
Two types of tests can be done for detection of sulphur.
• Lead acetate test :
Sodium extract is acidified with acetic acid solution and 1,2 drops of lead acetate solution is added to little sodium extract . Formation of black ppt. indicates the presence of sulphur in the organic compound.

• Sodium nitroprusside test :
Few drops of sodium nitroprusside solution is added to little sodium extract. Appearance of violet colour indicates the presence of sulphur in the organic compound.

Detection of both nitrogen and sulphur
If both nitrogen and sulphur are present in organic sample, sodium extract has following composition:

To this sodium extract, ferric chloride (FeCl3) solution is added, appearance of blood red colour indicates the presence of both N and S.

Detection of halogen
If halogens are present in organic sample, sodium extract has following composition :

A little sodium extract is taken in a test tube, few drops of nitric acid is added, boiled, cooled and finally few drops of silver nitrate solution is added.
1. If chlorine is present in the organic compound, white ppt. is obtained which is soluble in ammonia solution (i.e. NH4OH) and reappears on adding HNO3.

2. If bromine is present in organic compound, pale yellow ppt. is obtained which is partially soluble in NH4OH.
![]()
3. If iodine is present in the organic compound, dark yellow ppt. is obtained which is insoluble in NH4OH.
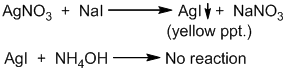
Q) Why Lassaigne’s extract is boiled with concentrated nitric acid while testing for halogen ?
Sometimes the organic compounds may contain N and S along with halogens.
If nitrogen is present in the organic compound, NaCN is formed in the sodium extract, which also gives white ppt. with silver nitrate solution.
![]()
If sulphur is present in the organic compound, Na2S is formed in the sodium extract which gives black ppt. with silver nitrate solution which masks the other colour.
![]()
Hence, before carrying out the test for halogens, the sodium extract is boiled with conc. HNO3 in order to remove cyanide(CN–) and sulphide (S – – ) as their volatile hydrogen acids, otherwise they would produce their own precipitate with AgNO3 and interfere with the result.
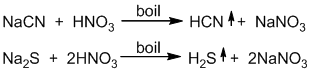
Objective Questions
1. In sodium extract solution, nitrogen of an organic compound is converted into :
a. Sodalime c. Sodium nitrate
b. Sodamide d. Sodium cyanide
2. During the detection of organic compound sodium extract is prepared to
a. Dissolve it in water
b. Convert it into ionic form
c. Make it more reactive
d. Make the reaction slow
3. The Prussian blue colour is formed in Lassaigne’s test if the nitrogen is present in an organic compound, the prussian blue colour is due to the formation of :
a. Na4[Fe(CN)6] c. Fe(OH)3
b. Fe4[Fe(CN)6]3 d. Fe4[Fe(CN)6]
4. Lassaigne’s test is not applied for the detection of :
a. Sulphur c. Nitrogen
b. Halogens d. Phosphorus
5. In Lassaigne’s test, if both N and S are present, blood red colour is seen due to formation of :
a. Ferric thiocyanate c. Ferric sulphide
b. Ferric cyanide d. Ferric ferrocyanate
6. Sodium extract is heated with conc. HNO3 before testing for halogens because :
a. Silver halides are insoluble in water.
b. Ag2S is soluble in HNO3
c. AgCN is soluble in HNO3
d. Na2S and NaCN are decomposed by conc. HNO3.
7. In Lassaigne’s test, if sulphur is present in organic sample then acidified sodium extract forms ____ ppt. with lead acetate solution.
a. Prussian blue c. Black
b. Blood red d. Violet
Answer :
1 – d, 2 – b, 3 – b, 4 – d
5 – a (also called- Ferric sulphocyanide)
6 – d, 7 – c
References
- Ghosh, S.K., Advanced General Organic Chemistry, Second Edition, New Central Book Agency Pvt. Ltd., Kolkatta, 2007.
- Morrison, R.T. , Boyd, R.N., Organic Chemistry, Sixth edition, Prentice-Hall of India Pvt. Ltd., 2008.
- March, j., Advanced Organic Chemistry, Fourth edition, Wiley Eastern Ltd. India, 2005.
- Sthapit, M.K., Pradhananga, R.R., Foundations of Chemistry, Vol 1 and 2, Fourth edition, Taleju Prakashan, 2005.
- https://www.pinterest.com/pin/834291899699006381/
- https://www.coursehero.com/file/29198433/Detection-of-foreign-elements-Copydoc/



