Contents
Laboratory preparation of Hydrogen sulphide (H2S)
Hydrogen sulphide (H2S) gas is prepared in the laboratory by treating iron sulphide with dil. H2SO4.
![]()
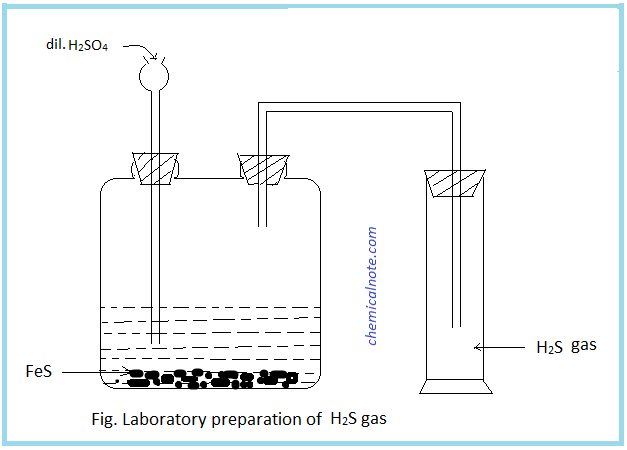
Procedure : Iron sulphide (FeS) pieces are kept in woulf’s bottle. Then a thistle funnel and delivery tube is fitted as shown in fig. When dilute H2SO4 is poured in woulf ’s bottle, reaction between FeS and dil. H2SO4 takes place and H2S gas is evolved which is collected in the gas jar by upward displacement of air.
Test of H2S : When a lead acetate paper is dipped in the jar containing H2S gas, it turns to black due to formation of lead sulphide.
![]()
Intermittent supply of H2S gas by Kipp’s apparatus :
(Working principle of Kipp’s apparatus )
{Intermittent = Stopping and starting at irregular intervals}
H2S gas is frequently required for salt analysis in the laboratory. The special apparatus which is used for the preparation of H2S is called Kipp’s apparatus.
![]()
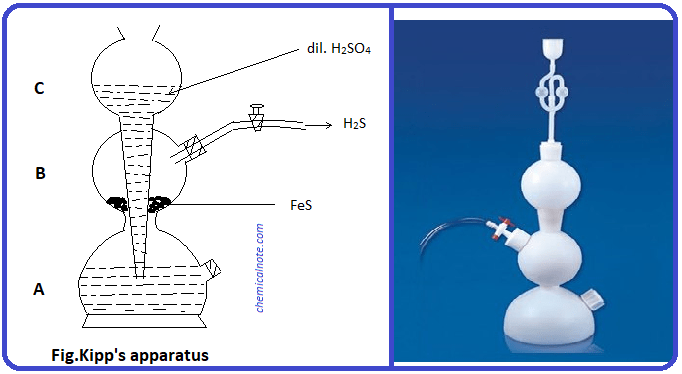
Kipp’s apparatus consist of three bulbs A, B and C. The bulbs are interconnected with each other. The upper bulb C contains a long stem which reaches upto bulb A. When dil. H2SO4 is poured from bulb C, liquid level rises in bulb A and reaches upto bulb B just to cover iron sulphide. Then the reaction between iron sulphide and H2SO4 takes place to form H2S gas. As H2S gas is formed, the pressure inside bulb B increases and forces the dil. H2SO4 up into the upper bulb C. Then the contact between FeS and dil. H2SO4 breaks and the formation of gas is stopped (ceased). When the tap is opened H2S comes out and the pressure decreases. As a result liquid level again rises at bulb B and H2S gas start to evolve again. In this way H2S gas is prepared intermittently by Kipp’s apparatus.
Physical Properties of H2S :
- It is colourless gas with rotten egg smell.
- It is soluble in water.
- It is poisonous gas.
{In its low concentration it causes nausea, dizziness and headache. At high concentration it is fatal (i.e. deadly poisonous). Conc. of H2S in air 1 part in 1000 parts of air may be fatal.} - It is heavier than air{with vapour density 17}
Chemical Properties of H2S :
1. Acidic character of H2S :
It acts as a weak diprotic (dibasic) acid. It ionizes in two steps to give two protons.

- H2S reacts with base to form two series of salts. Eg.

- H2S reacts with basic oxides to give salt.

2. H2S as reducing agent :-
Sulphur in H2S bears -2 oxidation state which in minimum oxidation state of sulphur. In chemical reaction, H2S tends to get oxidized . So H2S acts as reducing agent.
- It reduces halogens into halogen acids.

- It reduces sulphur dioxide to sulphur.
![]()
- It reduces ferric salt to ferrous salt.

- It reduces conc. HNO3 to NO2.

- It reduces conc. H2SO4 to SO2.

- It reduces acidified potassium permanganate solution and pink colour of KMnO4 is discharged.

- It reduces acidified K2Cr2O7 solution and orange color of K2Cr2O7 changes to green.

3. H2S as analytical reagent in salt analysis ( Precipitation of metal sulphide) :
H2S gas is used as analytical agent to detect group II basic radicals in acidic medium i.e. HCl and group IIIB basic radicals in in alkaline medium i.e. NH4Cl and NH4OH.
Group II basic radicals : Group II basic radicals consists of Cu ++ , Hg ++, Pb++, Cd++, Sn++, As+++, Sb+++, Bi+++. In qualitative salt analysis this group II radicals are precipitated in the form of their sulphide in acidic medium. By noting the colour of ppt., we can identify the metal cations.
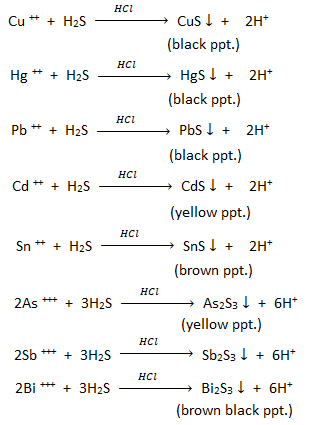
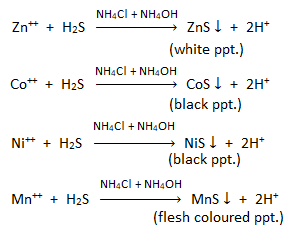
Group IIIB basic radicals : Group IIIB basic radicals consist of Zn++, Co++, Ni++, Mn++. These are precipitated by passing H2S in alkaline medium ( i.e. NH4Cl + NH4OH).
Structure of H2S :

Uses of Hydrogen sulphide (H2S) :
- It is used as analytical reagent in laboratory.
- It is used as reducing agent.
- It is used to prepare metallic sulphide, which can be used as pigment.
References :
- Shriver, D. F., Atkins, P. W., Inorganic Chemistry, Fifth Edition, Oxford university Press, 2010.
- Agrawal, S. K., Lal, K., Advanced Inorganic Chemistry, Fifth Revised Edition, Pragati Prakashan, Meerut, 2001.
- Cotton, F. A., and Wilkinson, G., Advanced Inorganic Chemistry, Fifth edition, John Wily and Sons, Singapore, 1995.
- Day, C.M., Selbin, J., Theoritical inorganic Chemistry, second edition, Affiliated East-West Press Pvt. Ltd., New Delhi, 2002.
- Mitra, L.A. , A Text Book of Inorganic Chemistry, Ghos and Company, 61st edition, 1996.
- https://en.wikipedia.org/wiki/Kipp%27s_apparatus
- https://www2.humboldt.edu/scimus/HSC.54-70/Descriptions/Kipp%27sGasApp.htm