Symbol : H
Atomic Number : 1
Atomic weight : 1.008
Electronic configuration : 1s1
Discovered by : Henry Cavendish
Henry Cavendish discovered hydrogen by passing steam through a red hot gun barrel (mostly iron)
-
He called it ‘inflammable air’. It’s name hydrogen was proposed by Lavoiser (Father of modern chem) as it produces water during burning with oxygen( Greek, hydro= water, gen= producer)
-
It is the most abundant and lightest element in the universe { and tenth most abundant element in the earth’s crust}
Contents
Position of hydrogen in the periodic table

It is the first element in the periodic table. It resembles the alkali metals (group IA) as it can be oxidized to H+ ion. On the other hand, it resembles the halogens (group VIIA) as it forms the hydride ion (H–). Hence ,the position of hydrogen in the periodic table is controversial or anomalous. Hence, it has been given a separate and distinct place unassociated with any particular group as the first element on left side of the periodic table on the top of the alkali metals (gr.IA).
Different forms of hydrogen
- Molecular hydrogen
- Atomic hydrogen
- Nascent hydrogen
- {Occluded hydrogen}
1. Molecular hydrogen
Ordinary hydrogen gas which consists of diatomic molecules in which two H atoms are combined together by a covalent bond is known as molecular hydrogen. Due to high H-H bond energy ( 104 KCalmol-1), molecules of hydrogen is very stable. It does not reacts under ordinary conditions.
2. Nascent hydrogen
(nascent=newly born) : The atomic hydrogen which is produced during the chemical reaction is called nascent hydrogen.
It is more reactive than ordinary hydrogen molecule. Thus nascent hydrogen is powerful reducing agent than ordinary hydrogen. Following reactions support this statement:
i. Reduction of potassium permanganate:
When molecular hydrogen gas is passed through acidified KMnO4, the pink colour of KMnO4 does not change.
![]()
But when a piece of zinc is treated with acidified KMnO4, it’s pink colour is discharged. This is because nascent hydrogen produced from the reaction between zinc and H2SO4 reduces KMnO4 into MnSO4 salt.
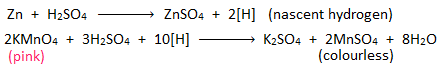
ii.Reduction of potassium dichromate:
When molecular hydrogen gas is passed through acidified K2Cr2O7, the orange colour of K2Cr2O7 does not change.
![]()
But when a piece of zinc is treated with acidified K2Cr2O7, it’s orange colour changes to light green. This is because nascent hydrogen produced from the reaction between zinc and H2SO4 reduces K2Cr2O7 into Cr2(SO4)3 salt.
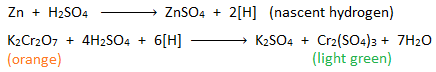
iii.Reduction of ferric chloride:
When molecular hydrogen gas is passed through ferric chloride, the yellowish brown(orange) colour of ferric chloride does not change.
![]()
But when a piece of zinc is treated with acidified FeCl3, its colour changes to pale green.

Uses : It is used as reducing agent.
3. Atomic hydrogen
It is hydrogen atom formed by dissociation of molecular hydrogen at about 1000 to 2000oC at low pressure. {This can be done by passing hydrogen gas through an electric arcstruck between two tungsten electrodes}

It has very short half life time of 0.3 second. Thus it is unstable.
It is more powerful reducing agent than molecular hydrogen and nascent hydrogen. It can reduce oxides and chlorides of metals like copper, silver, mercury to metallic form.

Uses of atomic hydrogen:
i. It is used as reducing agent.
ii. It is used as atomic hydrogen torch for welding metals of very high melting point.
Differences between nascent and atomic hydrogen :
| Nascent hydrogen | Atomic hydrogen |
| 1. Nascent hydrogen is formed in the reaction mixture at room temperature. |
1. Atomic hydrogen is produced at very high temperature of about 2000oC by dissociating molecular hydrogen. |
| 2. Nascent hydrogen is formed in situ but not been isolated yet. | 2. Atomic hydrogen can be isolated and has a half life of 0.3 seconds. |
| 3. It is weaker reducing agent than atomic hydrogen . It can not reduce certain metallic salts like AgCl, CuO, etc. | 3. It is stronger reducing agent than nascent hydrogen. It can reduce such metallic salts. Eg. |
4. Occluded hydrogen :
Adsorption of any gas on metal surface is known as occlusion. When molecular hydrogen is adsorbed on a metal surface (such as Fe, Au, Pt and Pd) , then this hydrogen is known as occluded hydrogen. In the powdered state, Pd can occlude approximately 1000 times its own volume of hydrogen. The occluded gas interacts with the metal to form a solid solution; or sometimes a part of the gas combines with the metal to yield other chemical compounds, for example, hydrides.
Ortho and Para hydrogen
Hydrogen molecule in which two hydrogen atoms have same nuclear spin (parallel nucleus spin) is called ortho hydrogen and hydrogen molecule in which two hydrogen atoms have opposite nuclear spin( anti-parallel nucleus spin) is called para hydrogen.
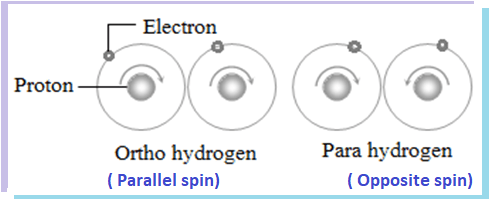
Therefore, in ortho hydrogen two nuclei spin in the same direction and resultant nuclear spin is high but in para hydrogen, two nuclei spin in opposite direction and the resultant nuclear spin is zero. Hence, para hydrogen has lower internal energy than the ortho hydrogen.
-
Due to difference in internal energy, the two forms of hydrogen have different physical properties like boiling point, specific heat, thermal conductivity, etc.
-
Since, para hydrogen has lower energy, at absolute zero, there is 100% para hydrogen and with increase in temp., proportion of para form decreases. At room temperature, hydrogen gas is mixture of 25% of para and 75% of ortho hydrogen.
Differences between ortho and para hydrogen :
| Ortho hydrogen | Para hydrogen |
|
|
|
|
|
|
|
|
Isotopes of Hydrogen
Isotopes are the atoms of the same element having same atomic number but different mass number. There are three isotopes of hydrogen:
- Protium or ordinary hydrogen
- Deuterium or heavy hydrogen
- Tritium or radioactive hydrogen.

| Name | Protium | Deuterium | Tritium |
| Symbol | 1H or H | 2H or D | 3H or T |
| No. of protons(P) | 1 | 1 | 1 |
| No. of neutrons(n) | 0 | 1 | 2 |
| No. of electrons(e) | 1 | 1 | 1 |
| Atomic no.(Z) | 1 | 1 | 1 |
| Mass no.(A) | 1.007 | 2.0014 | 3.017 |
Naturally occurring hydrogen contains about 99.985% of protium, 0.014% of deuterium and 0.001 % of tritium.
-
Isotopes have different physical properties since they differ in their mass number.
-
They have same chemical properties since their electronic configuration is same. However, they differ in the rate of chemical reaction. For example, D2 reacts with Cl2 about 13 times slower than H2 does. The different in rate of reaction due to difference in mass of the atoms of the same element is called isotope effect.
Uses of protium :
- It is used in filling {toy} balloons.
- It is used in synthesis of ammonia by Haber’s process.
- It is used as rocket fuel.
- It is used for the hydrogenation of vegetable oil to form vanaspati ghee.

Uses of deuterium :
- It is used in nuclear fusion reaction to liberate large amount of heat which is used in hydrogen bomb.
![]()
- It { and its compounds } is used as tracer to determine the mechanism of reactions.
- It is used to prepare heavy water.
Uses of tritium :
- It is used in nuclear fusion reaction to liberate large amount of heat which is used in hydrogen bomb.
![]()
- It is used as radioactive tracer { in absorption of water by plant roots.}
- It is used as radioactive substance in cancer therapy.
Heavy water and its applications
- Chemically heavy water is deuterium oxide (D2O).
- Its molecular weight is 20 amu.
- Heavy water is colorless, odorless and tasteless.
- The ordinary water contains small quantity of heavy water.
- Heavy water is obtained by prolonged or repeatedly electrolysis of water.
- Boiling point = 101.420C and freezing point = 3.80C.
Uses of heavy water:
- It is used as a moderator in nuclear reactors because it slows down the fast moving neutrons and thus helps in controlling nuclear reactions.
- It is used as a tracer to study the mechanism of metabolic processes and many organic reactions.
- It is used to prepare heavy hydrogen.
- It is used in NMR (nuclear magnetic resonance) spectroscopy.
Application of hydrogen as fuel
Hydrogen is an excellent substitute for fossil fuels such as coal, petrol, diesel, etc. Hydrogen fuel cells are growing technology offering significant economic, environmental and energy efficiency benefits.
The operation of hydrogen fuel cells is similar to that of a battery.

The hydrogen fuel cell has two electrods- an anode and a cathode, which are separated by a membrane. Oxygen passes over the cathode and hydrogen passes over the anode. The hydrogen reacts with a catalyst on the anode which converts hydrogen into positively charged ions (H+) and negatively charged electrons (e–). The electrons flowing out of the cell are used as electrical energy to power an electric motor. The hydrogen ions then pass through the membrane to the cathode and combine with oxygen and the electrons to yield water.
2H2 (g) + O2 (g) → 2 H2O (l) + energy
Advantages of hydrogen fuel cells:
- It is pollution-free source of energy. Unlike gasoline and diesel powered vehicles, hydrogen fuel cells do not produce air pollutants.
- It reduces the dependence on petroleum imports as hydrogen can be demostically produced from various sources.
- Fuel cell vehicles powered by hydrogen do not produce greenhouse gas emissions.
Uses of hydrogen fuel cells:
NASA (National Aeronautics and Space Administration) is the primary user of hydrogen resources for its space program. NASA fuels the booster rockets of the space shuttle using liquid hydrogen and employs hydrogen batteries for electrical sources.
- Hydrogen fuel cells can power any portable devices like video recorder, cellular phone, laptop, etc. that uses batteries.
- Hydrogen fuel cells can be used in vehicles such as buses, cars etc. to replace combustion engines or even large batteries.
- Hydrogen fuel cells can be a clean and reliable source of power to cities and buildings.
Questions and their answers :
1. Which of the following is most reactive form of hydrogen?
a. Nascent hydrogen c. Ortho hydrogen
b. Atomic hydrogen d. Para hydrogen
2. Water gas is a mixture of :
a. H2O and CO2 c. H2O and CO
b. H2 and SO2 d. H2 and CO.
3. Which of the following is known as heavy hydrogen?
a. Protium c. tritium
b. Deuterium d. para hydrogen
4. Which of the following is known as radioactive hydrogen?
a. Protium c. tritium
b. Deuterium d. para hydrogen
5. At room temperature, ordinary hydrogen is the mixture of ortho and para hydrogen in the ratio of :
a. 1:3 c. 2:3
b. 3:1 d. 1:2
6. When a piece of zinc is added to acidified potassium dichromate solution, the color changes to:
a. Yellow c. sky blue
b. Pink d. light green
7. Least abundant isotope of hydrogen is:
a. Protium c. tritium
b. Deuterium d. Heavy hydrogen
8. The adsorption of hydrogen by metals is called:
a. Dehydrogenation c. Occlusion
b. Hydrogenation d. Absorption
9. Molecular mass of heavy water in amu is:
a. 14 c. 18
b. 16 d. 20
Answers :
1. b 2. d
3. b 4. c
5. a 6. d
7. c 8. c
9. d
Note : 8. The adsorption of hydrogen by metals (such as Fe,Au,Pt and Pd) is called occlusion. In the powdered state, Pd can occlude approximately 1000 times its own volume of hydrogen.
Q) Give a reaction which shows that water contains hydrogen.
→ When steam is passed through red hot iron, hydrogen gas is obtained.
![]()
Since, iron does not contain hydrogen, it must come from water. This reaction shows that water contains hydrogen.
Q) When seam is passed through red hot coke, a mixture of two gases is obtained. Name the gases and write the reaction involved.
→ When steam is passed through red hot coke, a mixture of CO and H2 gas is obtained.
![]()
These gases CO and H2 together are called water gas.
References
- Sthapit, M.K., Pradhananga, R.R., Foundations of Chemistry, Vol 1 and 2, Fourth edition, Taleju Prakashan, 2005.
- https://www.energy.gov/eere/fuelcells/hydrogen-fuel-basics
- https://www2.lbl.gov/abc/wallchart/chapters/02/3.html


![Electrochemistry – Class 11 [NEB] Chemistry](https://chemicalnote.com/wp-content/uploads/2021/11/aldo-keto-cover.png)

