Health authorities have been giving us two messages : there is no any drug to kill the corona virus and also wash your hands to stop the virus spreading. World is struggling to make a drug for the corona virus but a simple soap kills the virus ! This seems odd but washing our hands with soap and water is one of the most important things we can do right now to stop the spread of coronavirus.
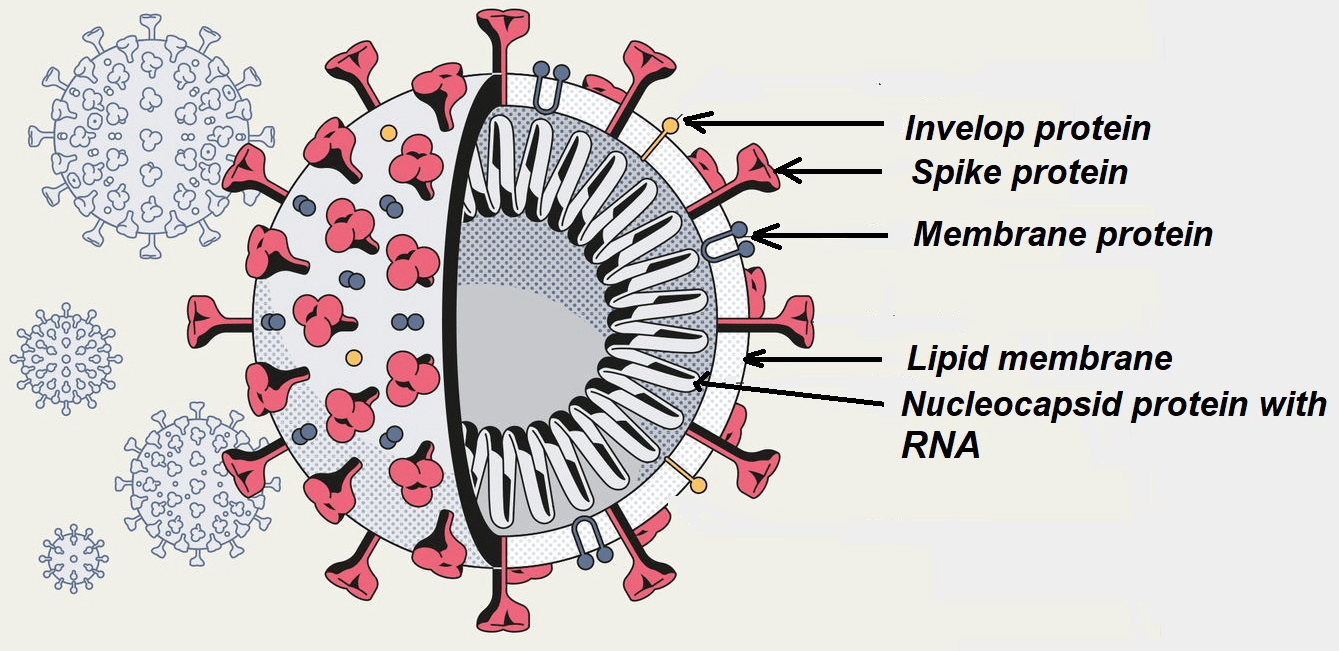
Most viruses consist of three key building blocks: RNA, proteins and lipids. The RNA, proteins and lipids self-assemble to form the virus. There are no strong covalent bonds holding these units together. A weak non-covalent interaction ( like hydrogen bonding and Vander- Waals attraction) is present between the proteins, RNA and lipids.
When a virus enters a cell (like human cell), it forces the cell to make fresh copies of its own RNA and the various proteins that make up the virus. These new RNA and protein molecules self-assemble with lipids (readily present in the cell) to form new copies of the virus. That means, the virus does not make new viruses; it makes copies of the building blocks, which then self-assemble into new viruses.
When an infected cell dies, all these new viruses escape out and go to infect other cells. In the lungs, viruses end up in the airways and mucous membranes. When you cough, or especially when you sneeze, tiny droplets from the airways can fly up to 10 meters. The larger ones are thought to be main coronavirus carriers, and they can go at least 2 meters. These tiny droplets end up on surfaces and dry out quickly. But the viruses are still active.
Now, chemistry says- similar molecules interact more strongly with each other than dissimilar ones. So, skin is an ideal surface for a virus. It is organic, of course, and the proteins and fatty acids in the dead cells on the surface interact with the virus . So, when you touch any substance with a virus particle on it, it will stick to your skin and, hence, get transferred on to your hands. If you then touch your face, specially your eyes, nose or mouth, virus can enter the body and you will be infected. Generally, people touch the face once every two to five minutes. So you’re at high risk once the virus gets on your hands, unless you wash off the active virus.

Washing the virus off with water alone might work. But water only is not enough at competing with the strong, glue-like interactions between the skin and the virus. Soapy water is totally different. People typically think of soap as gentle and soothing, but from the perspective of microorganisms, it is often extremely destructive. A drop of ordinary soap diluted in water is sufficient to rupture and kill many types of bacteria and viruses, including the new coronavirus that is currently circling the globe.
Hand soap is made up of molecules called surfactants. Surfactants have a hydrophilic (or water loving) end, and a lipophilic end (hydrophobic end), that likes lipids like those in the lipid layer of viruses, like coronavirus. When you wash your hands with soap and water, you surround all viruses present on your skin with soap molecules. Then the surfactants (soap molecules) can actually wedge their way into the lipid layer, lipophilic end first, and they break all the non-covalent interactions and the whole virus just falls apart like a house of cards (i.e. all building blocks of virus will break apart).
Surfactant molecules(soap) will come in and surround those broken down viral components and forms tiny bubbles called micelle . When you rinse your hands, the hydrophilic ends of the surfactants(soap molecules) will interact with water molecules and all the viruses that have been damaged, trapped and killed by soap molecules are washed away.




To be completely effective, make sure you’re really scrubbing with the soap, building up bubbles and getting all parts of your hands and under your nails for twenty seconds.
If you’re out and about, hand sanitizer with at least 60 percent alcohol can be used to sanitize your hands as well. Ethanol and other types of alcohol do not only readily form hydrogen bonds with the virus material but, as a solvent, are more lipophilic than water. Hence, alcohol does dissolve the lipid membrane and disrupt other non-covalent interactions in the virus.
However, you need a fairly high concentration (at least 60%) of the alcohol to get a rapid dissolution of the virus. Vodka or whiskey (usually 40% ethanol) won’t dissolve the virus as quickly. Overall, alcohol is not as good as soap at this task.
Disinfectants, or liquids, wipes, gels and creams containing alcohol (and soap) have a similar effect but are not as good as regular soap. Apart from alcohol and soap, antibacterial agents in those products don’t affect the virus structure much. Consequently, many antibacterial products are basically just an expensive version of soap in how they act on viruses. Soap is the best, but alcohol wipes are good when soap is not practical or handy.
REFERENCES :
- https://www.marketwatch.com/story/deadly-viruses-are-no-match-for-plain-old-soap-heres-the-science-behind-it-2020-03-08
- https://www.economist.com/briefing/2020/03/12/understanding-sars-cov-2-and-the-drugs-that-might-lessen-its-power
- https://www.theguardian.com/commentisfree/2020/mar/12/science-soap-kills-coronavirus-alcohol-based-disinfectants
- https://bearsvillesoaps.com/pages/why-soap-works
- https://www.irishtimes.com/life-and-style/health-family/why-soap-can-kill-coronavirus-and-how-its-discovery-changed-human-history-1.4210184


