Contents
Haloarenes (Aryl halides)
Aryl halides : The group of organic compounds in which the halogen atom is directly attached to the benzene ring.
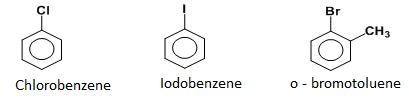
Aralkyl halides : The group of organic compounds in which halogen atom is linked to the carbon atom of the side chain.

Note: If benzene ring is taken as substituent then,
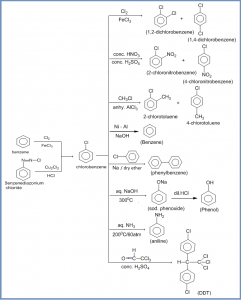
Preparation of chlorobenzene
1. By direct chlorination :
Chlorobenzene can be obtained by the direct chlorination of benzene in cold, dark and in the presence of halogen carrier Lewis acids such as AlCl3, FeCl3,etc. 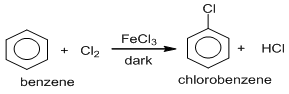
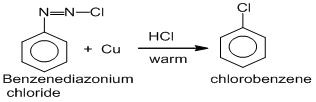
2. From benzene diazonium chloride :
Sandmeyer reaction :
Chlorobenzene can be prepared by treating benzene diazonium chloride with cuprous chloride dissolved in HCl. This reaction is called Sandmeyer reaction.

Gattermann reaction :
Chlorobenzene is prepared by treating benzene diazonium chloride with copper powder dissolved in HCl. This reaction is the modification of Sandmeyer’s reaction and called Gattermann reaction.
Q) What is diazotization reaction?
When aniline is treated with sodium nitrite (NaNO2) and dil.HCl at temperature below 50C, benzene diazonium chloride is obtained. This reaction is known as diazotization reaction.
Note : This is a reaction of aniline with nitrous acid but nitrous acid is unstable so it is prepared in situ by the reaction of sodium nitrite and dilute HCl.

Physical properties of haloarenes
1. Haloarenes are colourless liquid or crystalline solid.
2. Haloarenes are insoluble in water but soluble in organic solvents like benzene, ether, alcohol, etc.
3. Boiling and melting point : Haloarenes have higher boiling point than arenes of comparable molecular masses. This is due to the greater magnitude of Vander- Waal’s force developed by the polarity of C – X bond.
For the same aryl group, boiling point increases with the increase in size of halogen atom.

The boiling point of isomeric dihaloarenes are almost same but the melting point of para isomer is quite higher than that of ortho and meta isomer. This is due to its symmetrical structure which leads to close packing in the crystal lattice.

Chemical properties of haloarenes
1. Electrophilic substitution reaction of haloarenes
The resonating structures of haloarene are as follows:
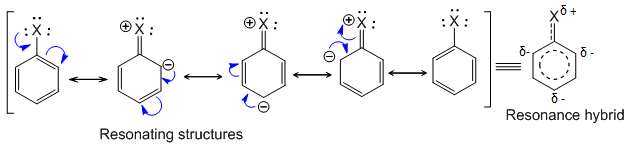
From the resonating structures it is clear that ortho and para positions are relatively rich in electron density and hence incoming electrophile attacks at these positions to give ortho and para substituted products. Hence, haloarenes (halogens) are o/p – directing towards electrophilic substitution reaction.
Note : Halogen shows –I effect. It withdraws electrons from benzene ring and ring gets somewhat deactivated as compared to benzene.
1. Halogenation : Halogenation takes place in the presence of iron or FeCl3 or anhydrous AlCl3 as a catalyst. Eg.

2. Nitration : Eg.

3. Sulphonation : Eg.

Note : p-product is major product and o-product is minor product.
4.Friedel-Craft alkylation : eg.

5. Friedel – Craft acylation : eg.

2. Reduction of haloarenes :
Haloarenes can be converted into the corresponding arenes by reduction with Ni-Al alloy in presence of alkali.

3. Reaction with sodium metal :
Fitting And Wurtz-Fittig reaction :
Haloarenes when treated with sodium in the presence of dry ether, diaryls are produced. This reaction is known as Fitting reaction.
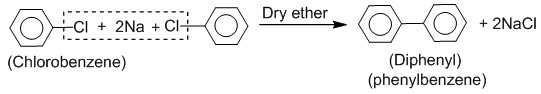
Haloarenes when treated with sodium and alkyl halide in presence of dry ether gives toluene. This reaction is known as Wurtz-Fittig reaction.

4.Nucleophilic substitution reaction in haloarenes :
Q) Why is it difficult to undergo nucleophilic substitution reaction in haloarenes?
In haloarenes electron pair of halogen atom is in conjugation with ∏ – electrons of benzene ring and following resonating structures are possible:

The resonating structures (II), (III) and (IV) indicates that C – X bond has partial double bond character. As a result, C – X bond in haloarenes is shorter and hence stronger as compared to the C – X bond of haloalkanes. Thus, cleavage of C – X bond becomes difficult which makes it difficult towards nucleophilic substitution reaction.
- However they undergo nucleophilic substitution reaction under drastic condition. Some of the examples of nucleophilic substitution reactions are :
1. Reaction with aq. NaOH : When chlorobenzene is heated with an aqueous solution of NaOH at 3000C and 200 atm gives sodium phenoxide which on acidification gives phenol.

2. Reaction with aq. NH3 : Chlorobenzene when heated with aq. ammonia in the presence of cuprous oxide (Cu2O) at 2000C under a pressure of 60 atm gives aniline.

3. Action with cuprous cyanide : Chlorobenzene when heated with cuprous cyanide (CuCN) at 2000Cin the presence of pyridine gives phenyl cyanide.

Phenyl cyanide can be used to prepare different important compounds such as:

5. Preparation of DDT :
Reaction of chlorobenzene with chloral :
DDT (Dichloro-diphenyl trichloroethane) is prepared by heating chlorobenzene with chloral in the presence of conc. H2SO4 (as the dehydrating agent).

Uses of DDT : It is used as a powerful insecticide to kill mosquitoes and other insects.
Haloarene (chlorobenzene) reaction practice chart
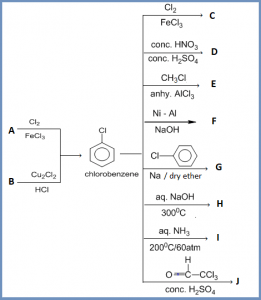
Answer :
Exercise
1. Which of the following reactions is wrong? Give reason.

2. An aromatic compound ‘A’ is formed by action of benzene with Cl2 in presence of FeCl3. When compound ‘A’ is heated with chloral in presence of conc. H2SO4 forms an insecticide ‘B’ used in agriculture. The compound ‘A’ when treated with Na in presence of dry ether gives ‘C’
a. Identify A, B and C. Write the reaction involved.
b. Reaction of aq.NaOH with ‘A’ is more difficult than with chloroethane. Justify with suitable example.
c. Write one use of B.
3. An organic liquid (A) reacts with sodium nitrite and dilute HCl in ice bath to give (B) which on warming with cuprous chloride in dilute HCl gives (C). When (C) is heated with NaOH at 3500C under high pressure followed by acidification ,(D) is formed. (D) reacts with aqueous bromine to give white ppt. (E) which on heating with Zn dust gives 1,3,5-tribromobenzene. Identify A, B,C, D and E with necessary chemical reactions.
4. Benzene diazonium chloride (BDC) is prepared by reacting primary aromatic amine with nitrous acid in ice-cold solution.
a. Write the chemical reaction that takes place in the preparation of BDC.
b. Write the uses of BDC in the Sandmeyer and Gattermann reaction.
c. What happens when diazotized solution of aniline is reacted with KI?
Write one example of each:
- Fittig reaction
- Wurtz-Fittig reaction
- Sandmeyer reaction
- Gattermann reaction
- Diazotization reaction
- Dow’s process
- Friedel-Crafts reaction
Convert:
- Benzene to benzoic acid
- Chlorobenzene to aniline
- Aniline to benzyl amine
- Nitrobenzene to bromobenzene
Give reason:
- Chlorobenzene gives ortho- and para- substituted product but not the meta- substituted product.
- Chloroalkane is hydrolyzed easily with aqueous NaOH but chlorobenzene requires high temperature and pressure.
- Melting point of p-dichlorobenzene is higher than that of o- and m-dichlorobenzene.
References
- Ghosh, S.K., Advanced General Organic Chemistry, Second Edition, New Central Book Agency Pvt. Ltd., Kolkatta, 2007.
- Morrison, R.T. , Boyd, R.N., Organic Chemistry, Sixth edition, Prentice-Hall of India Pvt. Ltd., 2008.
- https://socratic.org/questions/5873a0597c014930227865a2
- http://www.chemspider.com/Chemical-Structure.7676.html




