Contents
What are allotropes and allotropy?
The two or more forms of same element having similar chemical properties but different physical properties are called allotropes and the phenomenon is called allotropy. For example- diamond and graphite are two allotropes of carbon.
Allotropes of carbon, sulphur and phosphorus are described below.
Allotropes of carbon
Carbon exists on following allotropic forms:
 .
.
Crystalline form :
1. Diamond :

Diamond is the purest form of carbon. In diamond 1 carbon atom is bonded with 4 other carbon atoms by covalent bond forming tetrahedral structure. The bond angle between carbon atoms in diamond is found to be 109.50 and its bond length is 1.54 Å.
- It is transparent and sparks when light falls on it.
- It is the hardest substance known.
- It is purest form of carbon ( 100% carbon)
- It’s density is very high (3.5 at 150C)
- It has very high refractive index ( 2.417)
- Its melting point is very high (37500C).
- It is bad conductor of heat and electricity.
Note : Diamond is bad conductor because its electrons are not free to move due to the formation of sp3 hybrid covalent bonds between carbon atoms.
Uses of diamond :
- It is used in jewellery.
- It is used for cutting glass.

Note : Diamond is weighed in carat, 1 carat = 0.2 gm
2. Graphite :

In graphite one atom is attached with other three carbon atoms by covalent bond which forms hexagonal ring. Out of four valence electrons only three valence electrons are used to form three covalent bonds leaving one electron free. This free electron per atoms is responsible for making graphite a good conductor. The hexagonal rings of graphite form a layer structure. The two adjacent layers of graphite are held with each other by Vander Waal’s force of attraction. Distance between two layers is 3.4 Å. The bond length between two carbon atoms is 1.42 Å and bond angle is 1200.
- It is good conductor of heat and electricity.
- It’s melting point is 35000C.
- It is resistant towards many chemical substances but it burns on strong heating.
Uses of graphite :
- It can mark the paper. So, it is used to prepare lead of pencil.
- It is used to prepare electrodes.
- It is used for the preparation of artificial diamond.

Note : Graphite can be converted into diamond at about 16000C and 50,000 to 60,000 atm pressure. Because of high cost and poor quality, diamond is seldom (rarely) made artificially.
3. Fullerene :
It is latest discovered allotropic form of carbon (in 1985). The most common form of fullerene is C60. It is named as Buckminster fullerene after the name of Rechard Buckminster. It contains 60 carbon atoms arranged in hexagonal and pentagonal ring which forms spherical bucky ball or soccer like structure. So, C60 molecules are also called bucky balls.
- It is soluble in organic solvent.
- It is good conductor of electricity.
Note : Other forms of fullerene are C32 , C50 , C70 , C76 , C84 , etc.

Uses of fullerene :
- It is used to make superconductor (due to its reasonable electric conductivity).
- It is used to make carbon nanotubes.
- It is used as molecular sieve.

Amorphous forms :
1. Coal : It is black or almost black solid combustible substance. It is naturally formed by the partial decomposition of wood or vegetable matter in presence of moisture at high pressure and temperature. It is used as fuel.
2. Coke : It is the residue of coal obtained after destructive distillation of coal. During this process volatile organic substances escape out. It is pure form of carbon, used as fuel as it burns without smoke. It is also used as a reducing agent in metallurgy.
3. Charcoal : It is porous form of carbon produced by the destructive distillation of organic materials like wood, sugar, bone, etc. Organic materials are heated strongly in limited supply of air. Charcoals are highly porous and used as adsorbent for absorbing toxic gases and purifying liquids.
4. Gas carbon : Gas carbon is dense form of deposited carbon on the interior part of gas retort during the manufacture of coal gas. It is good conductor of electricity and is used for making electrode in dry cell.
5. Lamp black : Lamp black is a finely divided black powdered soot obtained by burning natural gas and other carbon rich compounds in limited supply of air. It is used in manufacture of ink, black paints, boot polishes, carbon paper, etc.


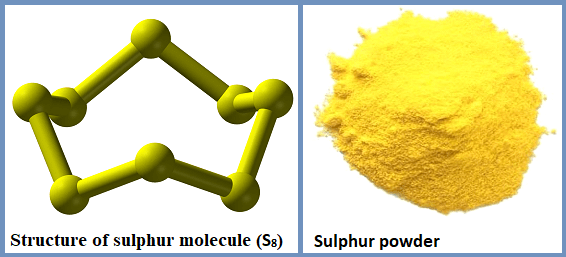
Allotropes of sulphur
Sulphur exists on following allotropic forms :

Crystalline Allotropes :
1. Rhombic sulphur ( α – sulphur ) :
- It is most stable form of sulphur at ordinary temperature.
- The crystals are octahedral (eight sided).
- It has pale yellow colour.
- It is insoluble in water but soluble in carbon disulphide (CS2).
- It’s melting point is 1140C.
- It is bad conductor of heat and electricity.
- Above 960C, the rhombic sulphur changes to monoclinic sulphur.

2. Monoclinic sulphur ( β – sulphur) :
- It is needle shaped crystalline sulphur.
- It is stable allotrope of sulphur above 960C.
- It is insoluble in water but soluble in carbon disulphide (CS2).
- It’s melting point is 1190C.

Note : 960C is the transition temperature at which both α and β forms can co-exist. Below 960C rhombic sulphur is stable and above 960C monoclinic sulphur is stable.

Amorphous allotropes :
1. Plastic sulphur ( γ – sulphur) :
- When boiling sulphur is poured into cold water, a rubber like substance is formed which is plastic sulphur.
- It is insoluble in water and CS2.
- It is super cooled liquid and does not have sharp melting point.
- On long standing, plastic sulphur changes to rhombic sulphur.
2. Colloidal sulphur ( δ – sulphur) :
It is prepared by passing H2S gas to the solution of oxidizing agents like SO2, HNO3, KMnO4, K2Cr2O7, etc.

- It is insoluble in water but soluble in CS2.
- It has no sharp melting point.
- On long standing or heating, it changes into rhombic sulphur.
3. Milk of sulphur :
- When sulphur is boiled with milk of lime [Ca(OH)2] then water soluble mixture of calcium pentasulphide (CaS5) and calcium thiosulphate (CaS2O3) is obtained. This mixture is then treated with dil. HCl and amorphous milk of sulphur is precipitated.

- It is white solid insoluble in water but soluble in CS2.
- It has no sharp melting point.
- It is bad conductor of heat and electricity.
- On heating it changes to rhombic sulphur.
Allotropes of phosphorus
Phosphorus exists on following allotropic forms :

Among them white and red phosphorus are more common.
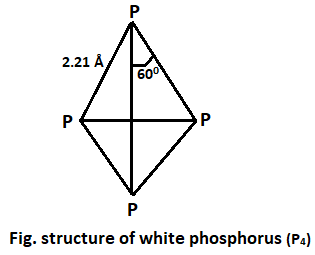
White Phosphorus :
White phosphorus is manufactured by heating calcium phosphate i.e. Ca3(PO4)2 with sand (SiO2) and coke (C) .
Calcium phosphate reacts with sand to form calcium silicate and phosphorus pentoxide.
![]()
Phosphorus pentoxide in vapour form is reduced by coke and liberates phosphorus at 15000C.
![]()
- It is soft, waxy white solid with garlic odour.
- Its melting point is 440C and boiling point is 2870C.
- It is extremely poisonous.
- Its ignition temperature is 350C. It burns (oxidize) in air giving yellow-green flame forming phosphorus pentoxide and trioxide. This phenomenon is called phosphorescence.
![]()
Hence, it is stored under water.
Uses of white phosphorus :
- It is to prepare poison for killing rats.
- It is used to prepare phosphine gas, phosphoric acid, etc.
- Hypophosphite prepared from white phosphorus is used in medicines as tonic.
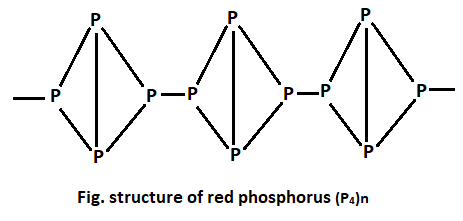
Red Phosphorus :
- When white phosphorus is heated at about 2500C in an inert atmosphere( of nitrogen or carbondioxide or coal gas) for several hours then it is converted into red phosphorus.

- It is dark red powder.
- It is colourless and non-poisonous.
- Its melting point is 5500C and sublimes at 2900C in absence of air.
- It is less reactive than white phosphorus.
- It does not show the phenomenon of phosphorescence.
Uses of red phosphorus :
- It is used in the manufacture of phosphate fertilizer.
- It is used for making match stick in match industry.
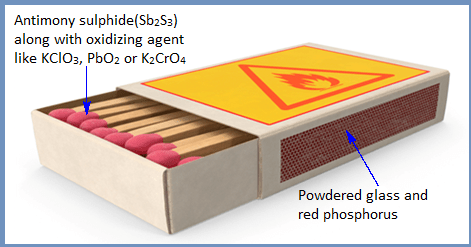
Note: The tip of match stick contains combustible material Sb2S3 along with oxidizing agent like KClO3, PbO2 or K2CrO4 and the two sides of the match box is coated with a mixture of powdered glass (abrasive) and red phosphorus. Friction applied to the side of the match box vaporizes red phosphorus and ignites. Thus produce fire to the head of match stick.
References
- Lee, j. D., Concise Inorganic Chemistry, Fifth Edition, Joh, Wiley and Sons, Inc., 2007.
- Sarkar, R., General and Inorganic Chemistry, Second Edition, New Central Book Agency(P) Ltd., India, 2007.
- Shriver, D. F., Atkins, P. W., Inorganic Chemistry, Fifth Edition, Oxford university Press, 2010.
- Agrawal, S. K., Lal, K., Advanced Inorganic Chemistry, Fifth Revised Edition, Pragati Prakashan, Meerut, 2001.
- Cotton, F. A., and Wilkinson, G., Advanced Inorganic Chemistry, Fifth edition, John Wily and Sons, Singapore, 1995.
- https://www.simply.science/images/content/chemistry/metals_and_non_metals/oxygen_family/conceptmap/Monoclinic_sulphur.html#:~:text=Monoclinic%20sulfur%20is%20a%20crystalline,ring%20molecules%20in%20crystalline%20structure.
- https://geology.com/minerals/graphite.shtml
- https://www.sciencedirect.com/topics/engineering/red-phosphorus


![Electrochemistry – Class 11 [NEB] Chemistry](https://chemicalnote.com/wp-content/uploads/2021/11/aldo-keto-cover.png)