Acetoacetic ester synthesis (of ketones)
Ethyl acetoacetate is called as acetoacetic ester.

Acetoacetic ester is an extremely useful molecule that can be used to prepare ketones and other molecules.
Synthesis of wide variety of organic compounds starting from acetoacetic ester is called acetoacetic ester synthesis.
Acetoacetic ester synthesis resembles with malonic ester synthesis and involves the following steps:
Step I : The α-hydrogen in acetoacetic ester is acidic in nature. When acetoacetic ester is treated with strong base like sodium ethoxide, it is converted into salt known as sodioacetoacetic ester.

Step-II : The carbanion thus produced is a nucleophile and attacks alkyl halide to form an alkylacetoacetic ester.

If required, the alkylation can be repeated to produce dialkyl acetoacetic ester.
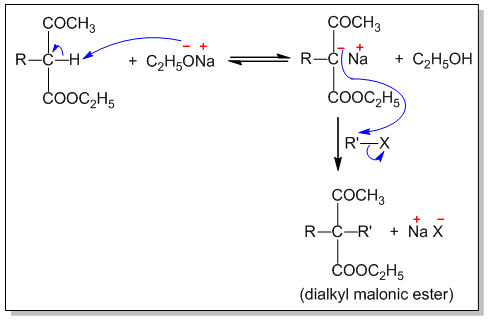
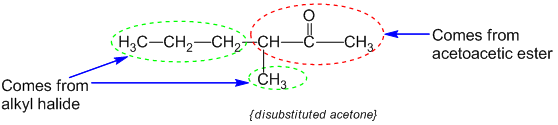
Step-III : These mono and dialkyl acetoacetic esters give the corresponding acids on hydrolysis. These acids undergo decarboxylation to form ketones.


Examples of acetoacetic ester synthesis
Q) Outline the synthesis of 3-methyl-2-pentanone from acetoacetic ester.
Structure of 3-methyl-2-pentanone is:

3-methyl-2-pentanone can be synthesized from acetoacetic ester by following reaction mechanism:

Q) Outline the synthesis of 3-methyl-2-hexanone from acetoacetic ester.
Hint:
Two alkyl halides used:
![]()
Q) Outline the synthesis of 5-methyl-2-hexanone from ethyl acetoacetate.
Structure of 5-methyl-2-hexanone is:

5-methyl-2-hexanone can be synthesized from acetoacetic ester by following reaction mechanism:

Q) Outline the synthesis of methyl isopropyl ketone from ethyl acetoacetate.
Hint:

Alkyl halides used:
CH3-Br two times.
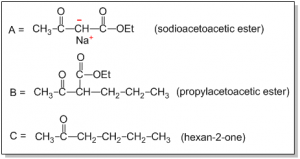
Q) What are A, B and C?

Ans:
References
- Ghosh, S.K., Advanced General Organic Chemistry, Second Edition, New Central Book Agency Pvt. Ltd., Kolkatta, 2007.
- Morrison, R.T. , Boyd, R.N., Organic Chemistry, Sixth edition, Prentice-Hall of India Pvt. Ltd., 2008.
- March, j., Advanced Organic Chemistry, Fourth edition, Wiley Eastern Ltd. India, 2005.
- https://chemicalnote.com/malonic-ester-synthesis-of-carboxylic-acids/